
A Wikiblog E-Book by Norman Uphoff with many others
Chapter 4: THE CONTRIBUTIONS OF ROOT SYSTEMS TO SRI SUCCESS
The first part of this memoire will reconstruct how a scientifically-validated understanding of the System of Rice Intensification emerged during the 25 years after Fr. Laulanié’s death, moving beyond what he had been able to assemble by the time of his death in 1995.[1] This first part of the SRI story gives an account of the diverse contributions made by researchers and others in many different countries to drawing a coherent picture of SRI. Ideas and evidence came from many sources, including farmers and lay persons, not just agricultural scientists.
The insights and information that helped to build up a comprehension of SRI are not presented here in chronological order because that would make the account rather hard to follow, given that this understanding was arrived at in an accretionary, unplanned manner. How much serendipity was involved in putting together the pieces of the SRI puzzle is an interesting feature of the story.
This part is organized in topical rather than sequential chapters, with a synopsis integrating the main explanations in a short appendix at the end. There was no research plan, and there were no formal hypotheses being tested, so the accumulation and synthesis of knowledge proceeded inductively, much as Laulanié had carried out his investigations that led to the assembly of SRI over a span of three decades. The inquiries and tests that were conducted were marked by curiosity and open-mindedness, often encountering the opposite of these two traits, as seen in Part II.
For most of those who studied SRI or aspects thereof, there was usually an underpinning of skepticism because SRI results were so much at odds with accepted plant and soil science doctrine. This skepticism was often accompanied by some enthusiasm, however, because the results being observed were so remarkable and beneficial. Enthusiasm is widely considered to be unscientific, or even anti-scientific. But since scientific issues are supposed to be resolved on the basis of evidence, investigators’ attitudes should be regarded as incidental, and not necessarily detrimental.
Skepticism is always appropriate for scientific investigation. But we learned in the course of studying SRI that skepticism is something to be optimized rather than maximized. Intransigent skepticism is alien to science when certain ways of thinking are clung to even when countervailing evidence becomes available.
UNDERSTANDING RICE PLANTS AS OPEN SYSTEMS RATHER THAN AS CLOSED SYSTEMS:
A FUNDAMENTAL INSIGHT
It was initially quite difficult to understand and accept Tefy Saina’s claim that with SRI methods, farmers could get rice yields of 5, 10 or even 15 tonnes per hectare -- on poor soils, without purchasing new inputs, and using the same varieties that farmers were already planting. Once this had been confirmed by three years of on-farm trials in and around Ranomafana, the question became: how was it possible to achieve a quadrupling of yield on these nutrient-poor, acidic soils, without introducing ‘improved’ varieties and without supplying compensatory chemical fertilizers? And applying less water? This was perplexing.
When I returned to Cornell after my first visit to Madagascar in 1993, I called my good friend and colleague Ronnie Coffman, who was a professor of plant breeding and a specialist on rice. For the 10 years before he joined Cornell’s faculty, Ronnie had coordinated the Asian rice research network of IRRI, the International Rice Research Institute in the Philippines. I relayed to him what I had been told about SRI by Tefy Saina to get his assessment.
Ronnie was understandably rather incredulous about what I reported to him. His first comment was that it would be very surprising if something so radically different from standard practice could be so successful. After all, a huge amount of research had been conducted on rice for many decades in many countries. Thousands of scientists had not hit upon such productive methods. He was particularly doubtful about the feasibility of attaining large increases in yield by increasing the number of tillers (stalks) per plant as was recommended by Tefy Saina.
Rice scientists agreed that having wider spacing between plants will increase the number of tillers put out by each plant. But it was also expected that having fewer plants per square meter would reduce the number of tillers per unit area, per square meter, and yield should be evaluated on an area basis, not per plant. Possibly having fewer plants per square meter, each with more tillers, could compensate for having lower plant density, but this was not the usual experience. (Later we learned that this was due to paddy soils being kept flooded.)
Rice scientists had found that when rice plants have a larger number of tillers, they invariably have a smaller number of grains per panicle.[2] Ronnie explained that when a rice plant puts out more tillers, which Tefy Saina was very pleased about, the number of grains per panicle will go down, an example of diminishing returns. This was the conclusion of scientific evaluations.[3] Even if the number of tillers per square meter was increased, the rate of increase in the total number of grains per square meter was bound to decline because tiller would each have fewer grains. These diminishing returns would cause the rate of increase in yield to decelerate rather than accelerate. Such large increases in yield as reported with SRI would require acceleration.
In economics, it is accepted that there is always some trade-off between and among the different factors of production. Getting more of one thing means getting less of another. This principle is expressed in the aphorism ‘there is no free lunch.’ Everything has its costs. It made sense that a rice plant which grew more tillers would have fewer grains per tiller.
When this relationship was explained to me, it made sense, and I accepted it. Yet I also knew that farmers around Ranomafana were quadrupling their yields of rice by using SRI methods. This was not possible if the inverse relationship proposed by rice scientists was correct, if rice plants that have more tillers have diminished grain formation and filling. This would make implausible the four-fold increase in yields that we were seeing and measuring.
In October 1998 when visiting Ranomafana, I was given a set of data collected by one of the four SRI trainers whom Tefy Saina had posted there, Simon-Pierre Rafanomezantsoa. He was Tefy Saina’s most experienced field worker, having worked closely with Fr. Laulanié for several years before the priest-agronomist died. Simon-Pierre was known to be hard-working and completely reliable, so the data that he had collected from 74 farmers who had practiced SRI that season in the Ambatovaky area on the western side of Ranomafana Park deserved to be taken seriously.
For each of these 74 farmers’ rice fields, Simon-Pierre using accepted sampling methodology had, along with other measurements, done counts of the number of tillers per plant and of the number of grains per panicle. Then he calculated the average number of tillers per plant and of grains per panicle for each farmer’s field.
During a flight from Antananarivo to Jakarta, I inspected Simon-Pierre’s data set and graphed by hand the relationship, shown from his data, between tiller number per plant and grains per panicle. To my surprise, instead of observing a negative correlation between these two variables -- the inverse relationship that rice scientists would predict -- there was the positive correlation seen in the figure below. Farmers whose methods produced more tillers per plant had also more grains per tiller.
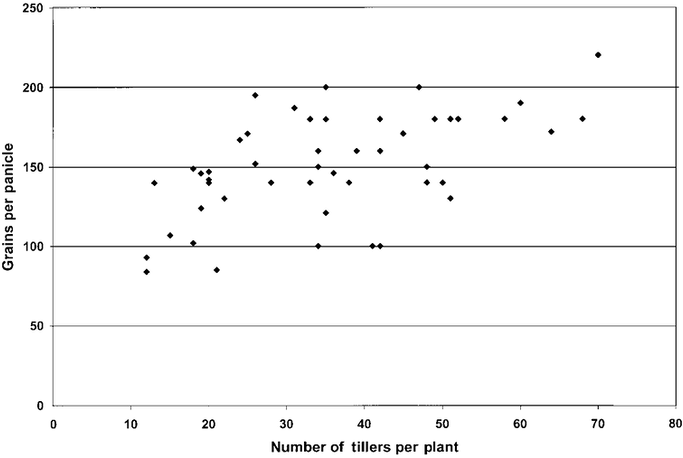
Number of grains per panicle associated with number of tillers per rice plant using SRI methods, Ambatovaky, 1997–98 season (N = 74)[4]
This graph showed that SRI-grown rice plants were different from rice plants grown from older seedlings with close crowding and continuous flooding. As discussed below, SRI plants have larger, deeper root systems than those on conventionally-grown plants, and their roots do not senesce in the soil for lack of oxygen. When rice plants’ roots die back, these plants become closed systems, losing their capacity to take up nutrients They will have to make trade-offs, using the carbohydrate building-blocks that the plants photosynthesize in their leaves either for plant metabolism, for growing more tillers, or to produce more grains on the panicles that have flowered and formed spikelets, which turned into grains.
SRI plants with deeper, healthier and longer-lived roots function as open systems, having more roots, more tillers, more leaves, and more grains. They are not constrained in the same way as are rice plants that have limited and limiting root systems. This was the first big advance in understanding how SRI methods can achieve greater growth and productivity.
Rice plants whose root systems degrade and suffocate due to hypoxia caused by continuous flooding of their soil become closed systems. By changing the growing environment below and above ground, SRI methods keep both the roots and leaves of rice plants from dying back (senescing) as occurs when rice plants are grown with crowding and flooding.
SRI rice plants with large and longer-lived root systems can continue to take up nutrients from the soil while their leaves convert these photosynthetically into carbohydrates throughout the rice plants’ life cycle. Such plants will be less reliant on the translocation of carbohydrates from the rest of the plant into its panicles for grain formation and filling. This translocation of nutrients is a crucial process for rice plants grown under flooded, crowded conditions with impaired root systems. However, translocation will be less necessary if the plant has a continuing supply of photosynthates due to persisting activity in its roots and tillers. Recall the pictures of SRI roots in Chapter 1, both their larger size and their lighter color.
That SRI-grown rice plants are different from ‘normal’ plants was made evident by research findings reported at the first international conference on SRI, held in China in 2002 and discussed in Chapter 8. By 2002, we could consider reports on SRI research and practice in 15 countries.[5] The research findings that commanded my attention were presented by Dr. Tao Longxing, a senior plant physiologist at the China National Rice Research Institute in Hangzhou.[6]
Tao grew a variety of hybrid rice variety simultaneously in different plots using either SRI or standard methods. He took samples from both sets of plants from the respective plots at different stages of their growth and weighed their above-ground plant organs: their grain-bearing panicles, their leaves, sheaths and tillers, as well as any senescing plant organs.
The figure below shows the respective cumulative average weights of these organs as the plants progressed through their reproductive phases, from panicle initiation (initial heading) through grain filling and full heading, to several stages of grain maturation, from milky rice to waxy rice (also called dough rice), and finally to yellow rice ready for harvest.

The most obvious difference between the two kinds of rice plants was the ballooning of panicle weight (purple) along with the greater growth of other plant parts. In the SRI plants, later onset of senescence and less yellowing of leaves and sheaths was also seen (yellow areas on the graph). The rice scientists who made these measurements at ‘China’s IRRI’ saw that SRI practices were producing a different and better kind of plant from rice seeds that had the same genetic potential.
Tao studied the growth of roots separately, evaluating two hybrid rice varieties under SRI management vs. traditional rice cultivation (TRC) methods. As seen below, with SRI methods there was both more root growth, and the roots grew deeper, by 10-15 cm, into the more aerobic soil. The dotted lines portray the downward root development with traditional cultivation (TRC), while the solid lines show SRI root growth to be both greater and deeper.
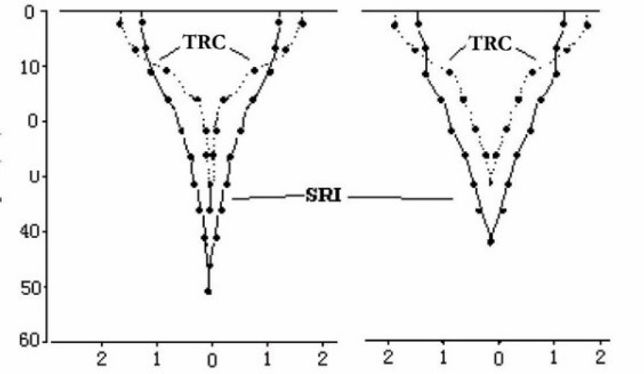
Xieyou 9308
Liangyou-peijiu
Under flooded conditions, the roots of conventionally-grown rice plants are known to remain mostly in the top 20 cm of soil.[7] With water abundant in the top layers of soil, and especially when fertilizer is applied to the soil, there is less need for the plant’s roots to search for water and nutrients in the soil’s lower horizons. Also, with less oxygen available, the roots have less metabolic ability to do this. With SRI management, on the other hand, rice plants have both more need and more capacity for vigorous root growth. This helped us understand how the growth and performance of rice plants’ roots played a major role in explaining SRI effects.
DEGRADATION OF ROOTS UNDER FLOODED SOIL CONDITIONS
In one of his unpublished technical papers, Laulanié commented on a lecture that he attended in 1991 at the Panorama Hotel in Antananarivo. This was given by a French agronomist Michel Puard on how rice plant roots degenerate for lack of oxygen when they are grown in flooded (hypoxic) soil. In his paper, Laulanié did not give details on that research, but in 1998, a Malagasy graduate student studying crop and soil science at Cornell, Solo Ranoromalala, helped me track down Puard’s published writings on this subject.[8] This made a huge contribution to our understanding of how SRI water management practices change the structure and performance of rice plants, particularly their roots.
Puard examined the roots of two varieties of rice, one bred for irrigated, flooded soil conditions (IRAT-173), and the other for upland, unirrigated conditions (IRAT-13). He compared cross-sections of their respective roots when each variety was grown in either saturated soil or in well-drained soil. The differences are seen below in the cross-sections of these two varieties’ roots when grown under different soil water conditions.
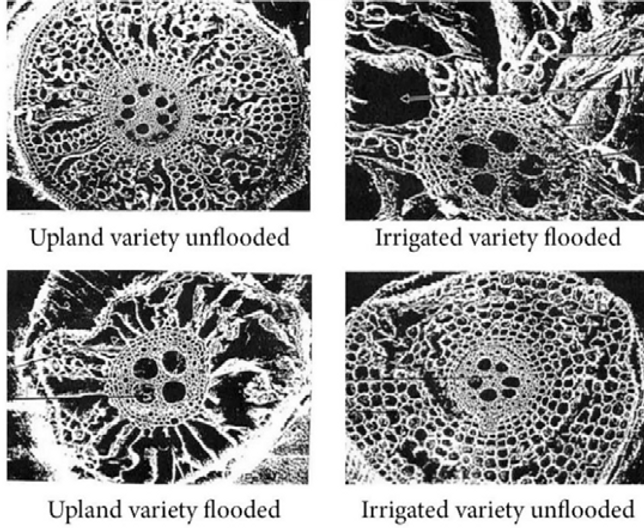
On the upper left, we see the internal structure of the roots of IRAT-13, the variety better for upland conditions, when grown in soil that was kept well-drained, not flooded. Its cross-section is symmetrical with the stele in the center, surrounded by cortical cells. The stele is the round body in the center of the root containing vascular tissues, the xylem that carries water and nutrients upward, and the phloem that conducts photosynthates from the leaves down into the roots. It looks very similar to the IRAT-173 cross-section seen in the lower right, when this variety doing well under irrigation is grown in unflooded soil. The pictures in the upper-left and lower-right both show the stele surrounded by regular and symmetrical cortex cells that store carbohydrates photosynthesized in the plant’s leaves. There is little evident difference between these two cross-sections.
What differentiates the two varieties is that when they are each grown under flooded conditions, the cortical cells that surround the stele of the IRAT-173 root (upper right) are more collapsed and irregular. This makes this variety better adapted to flooded soil conditions because its larger air-filled cavities (aerenchyma) allow more oxygen and other gases to diffuse in the roots through them. The more porous cortex of IRAT-173 plants helps to explain why this variety is more successful for irrigated production. The variety IRAT-13 which has less degradation of its cortical cells is better suited for rainfed cultivation with aerobic soil.[9]
It is widely and commonly believed that rice crops grow best when their fields are continuously flooded.[10] But this is a misconception. Flooding, to be sure, assures that the rice plants will always have water and thus can avoid the water shortages and stresses that commonly constrain the success of a rice crop that relies entirely on rainfall.
Flooding suppresses many weeds and thus reduces farmers’ labor requirements for weed control. But inundation also deprives rice plant roots (and beneficial aerobic soil organisms) of the oxygen that they need for their growth and survival.
Rice plants can form more and bigger aerenchyma in their roots than can other cereal crops like wheat and maize, so rice can survive in flooded conditions that are detrimental to these other crops. In fields that are flooded, rice can produce a good yield even if not its highest possible yield, whereas other crops would struggle just to stay alive.
The root cross-sections shown above suggest that rice is a species that evolved as an upland plant (compare the upper-left and lower-right pictures above). But some varieties of rice evolved the ability to form more and more effective aerenchyma in their roots. These air pockets could channel oxygen from the atmosphere into and through the roots, oxygen that would otherwise have had to be obtained through and from the soil.
Rice scientists, like farmers, have accepted the idea that it is natural for rice to be grown under flooded conditions, giving little thought to rice’s preference for well-drained but moist soil.[11] Rice crop breeders who sought to improve the production of irrigated rice selected genotypes that could form bigger and better air pockets in their roots.
In 1998 I happened to come across some research done in India 25 years earlier that assessed the effect of continuous flooding on rice roots, with trials done under carefully controlled conditions that permitted precise measurements.[12] Rice plants of the same variety were grown in pots with soil that was either continuously saturated or kept well-drained. The respective sets of roots were evaluated at successive stages to compare their growth.
The article’s abstract summarized the results as follows: “Root degeneration, a phenomenon normally unique to submerged conditions, increased with an advance in plant growth. At flowering [when the reproduction stage for producing grains begins], 78% [of the roots on rice plants under flooded conditions] had degenerated. During the first phase of growth under flooding, and throughout the growth period under unsaturated [soil] conditions, roots rarely degenerated.”
This research by Kar and associates, together with that of Puard and his French colleagues, could help to explain why SRI plants in Ranomafana, which were not kept continuously inundated, were so much more successful than rice plants of the same variety on the same (poor) soil. Being grown in soil that was alternately wetted and dried meant that the degeneration of plant roots was delayed and/or lessened. This has a profound effect of plants’ success as seen in the picture below from Indonesia, where Miyatty Jannah shows phenotypic differences between SRI and usual rice plants. Note especially the differences in root size and color.

However, the established view among rice scientists at the time was that rice is an aquatic plant which grows better under continuous inundation.[13] Rice specialists had paid little attention to the findings reported by Kar, Puard and others about root degradation resulting under hypoxic soil conditions. And the culture of continuous flooding of rice was well established in farming communities around the world, wherever enough water could be impounded, pumped or diverted for farmers to grow their rice in standing water. This wastes water and reduces rice yields by millions of tonnes each year.
TESTING ROOT-PULLING RESISTANCE
In 1997, before we knew about this research in France and India, we were able to get some agronomic and economic evaluations of SRI methods started in Ranomafana through cooperation with the Faculty of Agriculture (ESSA) at the University of Antananarivo. In March of that year, Glenn Lines, the in-country team leader for CIIFAD’s work on the project, and I visited the faculty’s director of research, Dr. Robert Randriamiharisoa, in his office at the university to solicit his assistance in studying SRI, to get some scientific understanding of what we were seeing.
Prof. Robert knew about SRI and was curious about it, even though the chair of the Agronomy Department, Dr. René Rabezandrina, was publicly antagonistic toward the new methods.[14] Prof. Robert’s contributions to a better understanding of SRI in the realm of microbiology are discussed in the next chapter, but here we note that he offered to assign the best graduating seniors in his Faculty of Agriculture to do their mandatory baccalaureate theses on SRI if our project would pay for their fieldwork expenses.
This was an offer to which Glenn and I quickly agreed. The first agronomy student whom Prof. Robert recruited to study the agronomic aspects of SRI was Joeli Barison, who turned out to be truly exceptional. (When Joeli graduated, he was awarded that year’s Laureate du Madagascar, the national prize given each year to the top university graduate in the whole country.) Robert said that he would personally help to develop the research design and would himself supervise the field research. Joeli’s agronomic study was complemented by the thesis research of a top economics student who evaluated the economic costs and benefits of SRI practices in Ranomafana.
Although neither Glenn nor I had done much thinking about roots, when we sat down to plan what we would like Joeli to examine, we thought that differences in root growth between SRI and conventionally-grown rice plants should be measured somehow. Assessing root characteristics and performance is challenging because roots are difficult to observe or measure underground. This is one reason why relatively little rigorous research has been done on plant roots.
We came up with a way to measure roots that, it turned out, had already been developed by IRRI scientists. It was called root-pulling resistance.[15] For this study we proposed using a simple market scale (with a spring mechanism). One end of a string would be tied around the base of a rice plant (or clump of rice plants) and the other end would be tied to the bottom of the scale. The top of the scale would then be tied to the middle of a length of bamboo. This would be held horizontally by two persons and slowly pulled upward from both ends while a third person watched the scale’s needle to see at what point (weight) the plant roots would be released from the soil. This would indicate how many kilograms of force were required to uproot the plant or plants.
This was, admittedly, a crude measure, and valid only if the soils in which the plants were growing would be similar in texture and moisture. However, it had the advantage of assessing the size and effectiveness of the whole root system, including the uncountable numbers of hair roots that extend out from the visible root structure. These are microscopic in size and vastly increase the surface area of the plant’s root system for absorbing water and nutrients. The whole root system is in contact with soil particles, and a measurement of root-pulling resistance (RPR) reflects how much contact and friction the whole root system has with the soil. When Joeli wrote up his research, he could say that this method of measurement been developed and validated by the International Rice Research Institute.
Joeli’s field research for his baccalaureate thesis was very ambitious. He persuaded two farmers in Ranomafana to cooperate in evaluating SRI methods comparing them with their current practice. On their fields, 171 trial plots were laid out, 2x2 meters each, each with a 50 cm margin around them to prevent the effects of water or nutrient diffusion from one plot to another. The two farms were at 610 and 1230 m elevation, respectively, with different types of soil and different climates, so that SRI management effects were evaluated under somewhat different conditions.
The farmers were promised that if their yields from SRI management were not improved enough over their usual level of production, they would be compensated for their forgone production. The yields were expected to increase by enough to compensate for the extra work. The trials were laid out, with three replications, according to Fisher block design to ensure randomness. As the SRI yields were all around 8 tonnes per hectare, there was much farmer satisfaction with their participation in the experimentation, and there was no request for payment for their cooperation.
The treatments evaluated, respectively and collectively, different combinations of age of seedlings, number of plants per hill, spacing between plants, water management, weeding, and soil amendments.[16] The results of factorial trials with so many factors are unavoidably complicated, but all of the hypotheses from Laulanié’s work were supported by Joeli’s research results, with strong statistical significance. In this chapter, we are most interested in the root results. I will switch here to an emic account.
In May 1998, I made one of my semi-annual visits to Ranomafana. The rice crop had been harvested, and Joeli had finished his data-gathering and was into data analysis. When we met in the project office, he was particularly excited to inform me about the results of his root-pulling measurements. With a sampling of 10 SRI-grown plants and 10 plants grown according to farmer practice, he had found that the latter (3 plants in a clump) required 28 kg of force to uproot, while single SRI plants required an average force of 54 kg for uprooting, almost six times more force per plant!
I remember telling Joeli that these were wonderful results, but they were ‘too good’ to be credible. Joeli’s smiling face turned grim. He was disheartened to think that I was not fully confident in his research skills and his integrity. This in turn made me feel badly, so I suggested that we do our own root-pulling in some SRI and non-SRI fields.
Joeli knew a farmer named Feno who had an SRI field about a kilometer away, so we walked there, with more tension and less joviality than our conversation had started with that morning. Feno’s field and that of his neighbor, who had not used SRI methods, had both been harvested recently so we could do no harm by pulling up their respective root stumps and didn’t seek permission. Joeli said it would surely be okay with Feno.
I was able to pull up half a dozen clumps of rice plants in the neighbor’s field. One hill had as many as 8 plants crowded together with sickly roots, all darkened and fairly small. When we moved five meters into Feno’s field, I found it impossible to pull up any of his clumps (single plants). I could feel there about twice as much resistance as I had felt in the neighbor’s field, which was consistent with Joeli’s measurements.
When a few years later Joeli did thesis research on SRI for a master’s degree in crop and soil sciences from Cornell, he again evaluated RPR in carefully-managed trial plots in Madagascar. He found that SRI plants had, on average, 7 times more per-plant resistance to uprooting than did the clumps of three farmer-practice rice plants, started from older seedlings and with continuous flooding.[17] Even though the farmer-practice trial plots had 3.3 times more rice plants per square meter, the SRI plots yielded 2.4 times more grain. Getting 8-fold higher grain production per plant under SRI management reflected the differences that Joeli had measured in the root systems.
ROOTS ARE NOT A ‘WASTE’
It should be evident that plants’ fates are influenced first and foremost by their roots. But this is not how crop scientists and plant breeders have viewed roots. Twice during the early years of our SRI story, the chair of Cornell’s Department of Crop and Soil Sciences, an excellent scientist and good friend, John Duxbury, told me with others listening that “roots are a waste.”[18]
Why? I asked. “Because they lower the plants’ Harvest Index,” was the explanation. This index is the ratio between a plant’s total biomass and how much edible crop it produces.[19] How much of what is produced in and by a crop plant can be consumed? That percentage is to be maximized. Anything but the edible portion of the plant is considered to be a waste of photosynthate. A high Harvest Index is seen as indicating greater plant efficiency in converting the resources of nutrients, soil, water and sunlight into product for human benefit. It became a kind of ‘holy grail’ for crop breeders.
There is a basic operational flaw in the Harvest Index, however, because when it is calculated, ‘total biomass’ refers only to the plant’s above-ground biomass. Plant roots are not dug up and weighed when figuring the index because that requires a lot more work than cutting and weighing the above-ground plant, and comparing that with the weight of the food produced.
This calculation considers plants as closed systems. The index assumes that when there is more root growth and/or more growth of the plants’ above-ground canopy, both of which are supported by the photosynthesis in the plant’s leaves, there will necessarily be less photosynthate available to fill the plant’s grains and thus produce more food for human consumption. This ignores what the roots can be doing along with the leaves to increase the total amount of photosynthesis that occurs, resulting in more carbohydrates to support all of the plant’s organs.
This zero-sum thinking is reasonable only if the plant is a closed system, however, having to allocate a fixed amount of photosynthate among the roots, the canopy, and the grains. To the extent that SRI rice plants are and remain open systems, because their roots do not deteriorate, they can have both more roots and more leaves. This will result in the plants having both more and even heavier grains.
Roots are not a waste if the carbohydrate that they receive from the leaves supports greater metabolism and activity in the roots which then continue to take up more nitrogen and other elements from the soil that support the synthesis of compounds which will build up all parts of the plant. Also, if the roots and leaves do not senesce, this process goes on for a longer period of time. The roots and leaves of flooded rice plants, on the other hand, experience root degeneration and the leaves atrophy.[20]
Part of the paradigm shift for rice production which complements the mental change of regarding plants as open rather than closed systems is to look at the growth of plants’ root systems as being responsive to environmental conditions around them. Not the result of genetic determinism where the DNA in plants’ cells is a blueprint producing a predetermined root system according to fixed genetic potentials. This is not true at all.
To use an analogy from sports, the plant’s genome is more like a playbook than a rulebook. In a playbook for American football, for example, different guidance is given for choosing plays depending on whether your team has possession of the football on its own 20-yard line or on that of its opponent and thus near to the goal, and depending on which team is ahead in the scoring, and by how much. Which play will be best to attempt is contingent on many factors and considerations. Plants’ genes are more like a playbook than a rulebook or a blueprint, to be followed mechanistically.
The concept of plasticity has gained appreciation in recent year as we understand better how plant phenotypes -- actual plants, not the potential plants embodied in a given genome -- reflect the interaction between the plant’s genetic potential (G) and its environment (E). This was made dramatically clear by data sent to me by Roland Bunch, an NGO leader in Central America who served on CIIFAD’s international advisory committee while I was the Institute’s director.[21]
In her classic book on tropical soil management, Dr. Ana Primavesi of Brazil reported research findings from Mexico on root plasticity that were quite remarkable. Dry matter production in maize plants’ shoots and roots was studied when the plants were grown hydroponically, not in soil but in nutrient-rich water solutions. This design enabled researchers to vary nutrient availability quite precisely to see how this affected the growth of roots and shoots. The experiment evaluated the effects on roots and shoots after the plants had been grown for 14 days in different concentrations of nutrients in the solution, and with the nutrients replenished with different frequency.[22] Some very large effects were observed.
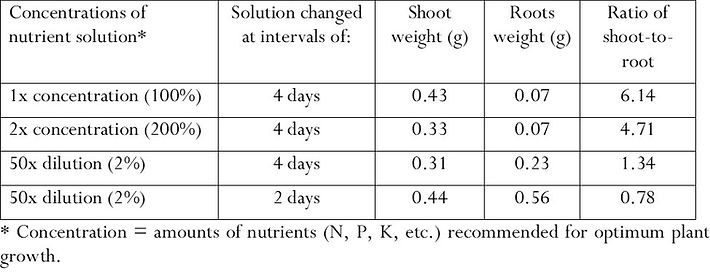
The first effect noted was that shoot biomass did not increase when the concentration of nutrients in the solution was doubled. In fact, supplying the plant with twice as many nutrients as plant nutritionists recommend for normal growth reduced shoot size by one-quarter -- and had no effect on the roots. To use an anthropomorphic expression, the roots could be lazy.
However, cutting the nutrient concentration to just 2% of the ‘normal’ concentration recommended for plant growth produced as much shoot growth as did the doubled concentration. But to exploit this diluted concentration, root growth more than tripled.
When this weak nutrient solution was changed (replenished) every other day, rather than every four days, the plants with a more continuous supply of nutrients at very low concentration were able to grow shoots as large as with a normal concentration. But to achieve this, there was an eight-fold increase in the weight of the roots that plants put out to exploit the limited but steady availability of nutrients.
These effects interested Roland because they could help to explain why compost applied to the soil, despite its lower volume of nutrients than contained in inorganic fertilizer, could produce comparable or better plant growth. He had learned about SRI from my CIIFAD reports and thought that this could be relevant to our search for explanations. While compost has lower nutrient content, its nutrients are released within the soil slowly and steadily as the organic matter is decomposed by microbial activity. (Roland’s work with SRI is described in his mini-memoire.)
For me, these data gave evidence of the kind of plasticity in root growth that we were seeing, the plant responding to soil conditions and its needs by growing larger and different root systems. This was evident in the pictures seen in Chapter 1. The SRI-grown rice plant in Cuba had a huge root system and 43 tillers, while the conventionally-grown plant had a much smaller and less vigorous root system, supporting just 5 tillers. The genetic potential of the two plants was the same as both were grown from the same variety of seed. However, their expression of that potential was radically different.
That the experimental evidence which Ana Primavesi reported and the visual evidence that we saw over and over again in SRI fields elicited so little interest from agronomists reflected their general neglect of roots. It was a sobering experience to consult the most widely cited textbook on rice.[23] In its 16-page index with over 1,100 entries, there was not a single entry on ‘roots’ to be found. There was one entry on ‘rhizosphere’ which referred to a sentence in the book which said that there is a rhizosphere. Nothing was said about its importance or function.
When I met the book’s author, S.K. De Datta, at a World Food Prize symposium reception in 1998, I mentioned to him my surprise that he had given so little attention to roots in his book. He in turn expressed his own surprise at this omission, seemingly unaware of this.[24] All of these pieces of evidence were eliciting a growing interest in me and in other SRI colleagues to understand better the contributions that root systems make to rice plants’ health and success.
DISCUSSING ROOTS AT THE INTERNATIONAL RICE RESEARCH INSTITUTE
More will be said in Part II about interactions with IRRI scientists regarding SRI, but giving here an account of my first discussion of roots with them moves the story along. In October 1998 when we were both attending the World Food Prize symposium in Des Moines, Iowa, I met the newly-appointed director-general of IRRI, Dr. Ron Cantrell, and informed him about what we had been learning about SRI in Madagascar, telling him that we would like to work with IRRI scientists in evaluating and understanding the effects that we were seeing in the field. He was very courteous and said that I would be welcome to give a seminar on SRI whenever I could visit IRRI’s headquarters in Los Baños, Philippines.
As it happened, the following February I was invited by the Asian Development Bank to participate in a workshop on participatory development being held in Manila, and Dr. Cantrell kindly arranged for an IRRI vehicle to pick me up at my hotel in the city and bring me to Los Baños, more than two hours away, for a presentation on SRI to IRRI staff.
There was a good turnout for the seminar, about 50 scientists and staff, and the questions after the presentation were mostly for clarification or elaboration, not taking much issue with what I reported.[25] Except for one slide which had showed pictures of rice root cross-sections from French research that were reproduced above for readers’ consideration.
My inference from these pictures -- that the formation of air pockets (aerenchyma) in rice roots had an inhibiting effect on roots’ uptake of water and nutrients -- was challenged by Dr. Guy Kirk, an IRRI specialist on nutrient uptake by roots. Guy said that the degeneration of cortical cells in rice roots to form air pockets did not have deleterious effects, citing research by Prof. Malcolm Drew at Texas A&M University. He noted that this research had been done on maize, but it should be relevant for rice as well.[26]
Upon returning to Cornell, I sent an email to Prof. Drew, reporting on my exchange with Guy Kirk. I attached a copy of my most recent paper on SRI and asked for references to his published papers so that I could read them and understand where I was wrong in my interpretation of Puard’s research in France.
Prof. Drew responded quickly, the next day, saying that he had read my paper overnight and found it very interesting. He wrote that in his research he had found that the formation of aerenchyma in plant roots did indeed have some negative effect on plants’ ability to take up water and nutrients. This response gave more confidence that we were on a reasonable track with SRI.
When I communicated Drew’s response to Guy Kirk at IRRI, he responded very graciously and collegially, sending me a number of papers on rice root research that he thought would be helpful to me as I continued working with SRI. These papers were indeed helpful because I would probably not have found them otherwise. Although I was not trained in agronomy, some of the findings reported were very clear and instructive.
One paper explained how, when soil is alternately flooded and dried, alternating back and forth between anaerobic and aerobic soil conditions, nitrogen (N), an essential nutrient for plants, will take the forms of both ammonium (NH₄⁺) and nitrate (NO₃⁻), with N attached to either hydrogen or oxygen and each molecule having a different electrical charge. In continuously flooded anaerobic soil, nitrogen is available mostly in the NH₄⁺ form, whereas in unflooded soil, NO₃⁻ is more prominent.[27]
The research showed that when the nitrogen in the soil is available in roughly equal amounts of NH₄⁺ and NO₃⁻, this contributes to higher grain yields than when the same amount of nitrogen is available in the NH₄⁺ form that predominates under flooded conditions. Thus, a water management regime that alternates wetting and drying of the paddy soil, as recommended for SRI, would give the rice plants more benefit from the same amount of N in the soil. This was something interesting and worth knowing.
Another paper reported that plant roots’ uptake of nitrogen is independent of the concentration of N at the roots’ surface. It is determined more by the plant’s internal condition. When rice plants have enough N to satisfy their internal needs, their roots down-regulate and reduce their uptake of N, even exuding surplus N that is not needed by the plant into the rhizosphere, the soil surrounding the roots.
Plants’ uptake of nitrogen is thus driven more by their demand for N than by the soil’s supply of N.[28] The Green Revolution approach was to ‘force-feed’ plants with large external supplies of inorganic nutrients, particularly nitrogen, to induce greater growth and more grain production. This puts the cart before the horse, however. It recalls the adage that ‘you can lead a horse to water, but you cannot make it drink.’
If rice plants are provided with a greater supply of N and other nutrients in the soil than they need, for reasons probably deriving from their evolutionary history, they will not use them unless their metabolism requires these nutrients, even returning excess nutrients to the soil. This could account, at least in part, for the disappointingly low uptake of N fertilizer by irrigated rice crops, usually only 20-35%.[29]
SRI management practices, on the other hand, planting younger seedlings widely spaced with no flooding that suffocates their roots, will induce more rapid growth of rice plants’ tillers and roots. This creates a strong plant demand for nitrogen. Realizing that plants’ demand for nutrients is more determinant of their uptake of nutrients than is their supply (subject of course to the constraint of availability) inverts the usual thinking about how best to manage plants’ nutrition and to accelerate their growth.
Of course, in agronomy a demand for nutrients will not necessarily create its own supply any more than supply creates its own demand in economics. This latter assertion, known in the economic literature as Say’s Law, has long ago been discredited. The Green Revolution strategy proceeded according to its own version of Say’s Law for nutrients, assuming that supply would create demand.
In crop management, much attention is given to the availability of nutrients. But nutrients are not an independent factor to be manipulated without regard to the other factors that contribute to plants’ growth and resilience. Success in agronomy requires an integrated approach, and SRI is a set of agronomic adjustments that promote rice plants’ demand for nutrients.
Probably the most surprising thing in the papers that I received from IRRI was a statement in a paper written eight years previously by Kirk with a Cornell agronomist colleague David Bouldin. They discussed in the article how aerenchyma (air pockets) are formed by the disintegration of about 30-40% of the cells in the cortex of rice roots, forming spaces within the roots through which oxygen can diffuse, eventually to the roots’ tips.
These authors stated that this disintegration “must surely impair the ability of the older part of the plant to take up nutrients and convey them to the stele.”[30] This wording was almost verbatim what I had said in my presentation at IRRI in March 1999. So why there was an objection raised to my talk remains obscure.
PAYING MORE ATTENTION TO THE GROWTH AND HEALTH OF ROOTS
The picture from Cuba seen in the first chapter, of huge root growth on Luis Romero’s SRI plant compared with that of its sister plant grown with his usual practices, was instructive. This root system seen on the right was more magnificent than usually seen even with SRI practices. But it showed the kind of growth that can be prompted by changes in how rice plants, soil, water and nutrients are managed. Its size was so great that it was hard to imagine how it could be explained just by differences in nutrient uptake and plant metabolism.
Understanding how rice plants could be induced to grow larger, healthier roots thus was an evident challenge. Their growth could be attributed to wider spacing between plants, and to increases in soil organic matter that improved the soil’s structure and made root growth easier. Also planting young seedlings enhances rice plants’ growth potential as discussed in Chapter 6. As reviewed in the next chapter, practices that promote the abundance and activity of microbial life in the soil are also a part of the explanation for SRI performance, as there is positive feedback between root growth and the associated populations of beneficial microbes. But getting farmers and scientists to inspect and assess the effects of SRI management on rice roots was important because the effects on roots can be so obvious.
We frequently received pictures from colleagues in various countries showing the differences in root growth that researchers and farmers saw when engaging in SRI cultivation. Below are pictures sent to Cornell by scientists in national rice research centers in Iran and Iraq. The dark coloration of the conventionally-grown rice plant roots in the left-hand picture shows the degeneration that Kar and his associates had documented with their controlled trials in the 1974 article that helped us begin understanding what was going on with SRI.[31] The size difference it very evident in both pictures.

When I first saw the picture sent by Rena Perez from Cuba, contrasting the two ‘sister’ plants, I began wondering: what else can be going on? The huge differences in the growth of rice plant roots was hardly explainable by the rampant unfolding of a genetic blueprint. As discussed in the next chapter, at least some of the answer might be found in the domain of soil biology, in the contributions that are made by beneficial soil organisms which live in the rhizosphere around the roots and which can also live inside the rice plants as symbiotic endophytes, a word meaning ‘inside the plant.’
In India, when SRI ideas and methods started to be extrapolated and applied to other crops like finger millet, wheat and mustard, some farmers and NGOs there renamed SRI as the System of Root Intensification.[32] This reflected the evident enhancement of root systems that resulted from starting with young seedlings (or direct seeding), wider spacing, active soil aeration when weeding, and increasing soil organic matter to improve soil structure and support diverse soil organisms. But this designation did not give attention and credit to the contributions made by these organisms, from earthworms to the tiniest of microbes. Understanding these biotic contributions became another major platform for our understanding how and why SRI management has the effects that it does, as discussed in the next chapter.
NOTES AND REFERENCES
[1] Laulanié’s understanding of SRI is summarized in his paper ‘Technical Presentation of the System of Rice Intensification, Based on Katayama’s Tillering Model.’ I translated this in 1998 with the help of Malagasy colleagues.
[2] Panicles are the heads or ears of grain that grow at the top of tillers which have flowered and become grain-bearing. They are also referred to as ‘fertile tillers’ because not every tiller (stalk) flowers and produces grains of rice.
[3] See, for example, J. Ying et al., S.B. Peng, Q. He, H. Yang, C. Yang, R.M. Visperas and K.G. Cassman, ‘Comparison of high-yield rice in tropical and subtropical environments: I: Determinants of grain and dry matter yields,’ Field Crops Research 57: 71-84 (1998). The ‘new plant type’ (NPT) that IRRI tried to create through plant breeding, to raise the presumed ‘yield ceiling’ in rice production was premised on having rice plants with fewer tillers but with all of them flowering and becoming fertile, and thus having more grains per panicle. G.S. Khush and S.B. Peng, ‘Breaking the yield frontier of rice,’ in: Increasing Yield Potential in Wheat: Breaking the Barriers, 36-51. International Center for Improvement of Wheat and Maize (CIMMYT), Mexico D.F., Mexico (1996).
It was not considered that tiller formation and root growth are coordinated processes, as both tillers and roots emerge concurrently from the same meristematic tissue at the base of the plant. This means that plants which have fewer tillers will also have fewer roots. This morphological coordination within rice plants doomed the NPT, which was a waste of millions of dollars of research funding.
[4] This graph is from Uphoff, ‘Agroecological implications of the System of Rice Intensification (SRI) in Madagascar,’ Environment, Development and Sustainability, 1: 297-313 (1999).
[5] The proceedings are on-line at: http://sri.cals.cornell.edu/proc1/index.html
[6] Tao Longxing, Wang Xi and Min Shaokai, ‘Physiological effects of SRI methods on the rice plant,’ in N. Uphoff et al., eds., Assessments of the System of Rice Intensification: Proceedings of an International Conference, Sanya, China, April 1-4, 2002, 132-136, CIIFAD, Ithaca, NY.
[7] A study of different field crops reported that 86% of flooded rice roots were in the top 20 cm compared to an average of 56% for five other field crops. J. Yamaguchi and A. Tanaka, ‘Quantitative observation on the root system of various crops growing in the field,’ Soil Science and Plant Nutrition, 36: 486-493 (2012).
In an earlier study, K.G. Cassman, S. Peng, D.C. Olk, J.K. Ladha, W. Reichert, A. Dobermann and U. Singh, ‘Opportunities for increased nitrogen use efficiency from improved resource management in irrigated rice systems,’ Field Crops Research, 56: 7-38 (1998), reported that 80-90% of rice roots are found in the top 20 cm of soil.
[8] This was before easy on-line computer searching. M. Puard, P. Couchat and G. Lasceve, ‘Importance de l'oxygenation des racines du riz (Oryza sativa) en culture inondée,’ L'Agronomie Tropicale, 41: 119-123 (1986) and ‘Etude des mécanismes d’adaptation du riz (Oryza sativa) aux contraintes de milieu. 1. Modification anatomique des racines. 2. Effets de la nutrition azotée sur la consummation d’oxygène par les raciness et l’evolution de l’acidité,’ L'Agronomie Tropicale, 44: 165-178 (1989).
[9] Subsequent research under experimental conditions showed that upland vs. lowland varieties of rice respond quite differently to hypoxia in terms of nutrient uptake. B.G. Zhang, M. Puard and P. Couchat, ‘Effect of hypoxia, acidity and nitrate on inorganic nutrition in rice plants,’ Plant Physiology and Biochemistry 28: 655-661 (1990).
[10] See endnote 13 below on this point.
[11] In October 1998, I happened to sit next to the late Dr. Nyle C. Brady, director-general of IRRI from 1973 to 1981, during a plenary of the World Food Prize symposium in Des Moines, Iowa. I had gotten to know him in the 1980s when I served on USAID’s Research Advisory Committee, which he chaired. When I told him about Puard’s research, which I had just read, and that by Kar et al., which is discussed next, he expressed surprise. His comment was that maybe at that stage of the rice plant’s growth, the plant did not need so many roots.
[12] It seems pure serendipity that I came across a reference to this article by S. Kar, S.B. Varade, T.K. Subramanyam and B.P. Ghildyal, ‘Nature and growth pattern of rice root system under submerged and unsaturated conditions,’ Il Riso, 23 (2): 173-179 (1974). This journal published in Italy is not widely known or read. Indeed, I had to procure it on inter-library loan from the University of Minnesota library as Cornell’s agricultural library, one of the best in the U.S., did not have it. By sheerest of coincidences, I had gotten acquainted previously with one of the co-authors, Dr. Ghidyal, a plant scientist, while he was serving as a Ford Foundation advisor in New Delhi in the 1980s.
[13] As noted in Chapter 1, one of the most authoritative books on rice stated categorically that rice “thrives on land that is water-saturated and even submerged, during part or all of its growth cycle” (page 43), and “Most varieties maintain better growth and produce higher grain yields when grown in flooded soil than when grown in unflooded soil” (pages 297-298). S.K. De Datta, Principles and Practices of Rice Production, Robert Krieger, Malabar, FL (1987).
[14] Professor René was vocally opposed to the promotion of SRI in Madagascar because he said that it was ‘backward’ and ‘too labor-intensive.’ He wanted Malagasy farmers to use what he considered ‘more modern methods.’ He particularly rejected the use of mechanical hand weeders which were recommended with SRI. He calculated and frequently spoke about how many kilometers a farmer would have to walk to weed one hectare of SRI paddy land with criss-crossing passes across the field. (Never mind that most rice farmers in Madagascar had fields that were only a small fraction of a hectare.) He advocated instead the use of herbicides to control weeds in rice paddies: ‘Herbicides better than manual weeding.’ We learned only later that Prof. René was a paid consultant for the German agrochemical company Hoechst, a conflict of interest that would have gotten him reprimanded in most universities.
[15] J.C. O’Toole and Soemartono, ‘Evaluation of a simple technique for characterizing rice root systems in relation to drought resistance,’ Euphytica, 30: 283-290 (1980).
[16] ‘Perspective de developpement de la region de Ranomafana: Les mechanisms physiologique du riz sur soils bas-fonds, cas de Systeme de Riziculture Intensive,’ Memoire fin d’etudes, Ecole Superieure des Sciences Agronomique, University of Antananarivo, Antananarivo (1998).
[17] J. Barison and N. Uphoff, ‘Rice yield and its relation to root growth and nutrient-use efficiency under SRI and conventional cultivation: An evaluation in Madagascar,’ Paddy and Water Environment, 9: 65-78 (2011).
[18] I had previously worked closely together on a large, multi-year sustainable agriculture proposal for USAID with John Duxbury, who served as chair of the Department from 1996 to 1999. In 1999, he conducted the first experimental trials of SRI in Nepal with unsatisfactory results because the trials were done during the monsoon season, and it was difficult to control water due to intensive rainfall. Planting tiny seedlings into deep-flooded fields would predictably fail since the protocol for SRI specifies that the soil should be well-drained or intermittently flooded and drained. Good results could not be expected if young SRI transplants were submerged.
Eventually John became a knowledgeable supporter of SRI modifications in rice practice, developing beneficial adaptions of SRI to raised-bed cultivation in Bangladesh. After visiting SRI fields in Nepal in 2006 with Rajendra Uprety, the first SRI ‘champion’ in that country, John became very interested in what more could be learned from SRI.
[19] http://www.dictionary.com/browse/harvest-index; ‘Harvest Index,’ section 6.4.1 of Plants in Action, textbook prepared for the Australian Society of Plant Scientists, New Zealand Society of Plant Biologists, and New Zealand Institute of Agricultural and Horticultural Sciences.
[20] I remember the excitement with which my Sri Lankan colleague and friend, C.M. Wijayaratna (Wijay for short) told me in 2001, after trying SRI methods on his family’s farm in the north of the country, that the flag leaf on SRI plants, the uppermost leaf that is the most photosynthetically active leaf on a rice plant, remained green for much longer than its counterpart on nearby conventionally-grown rice plants. Subsequently, many agronomic evaluations confirmed this observation.
[21] While he was Central America representative for World Neighbors, Roland authored a book that pioneered what is now known as ‘farmer-to-farmer extension’: Two Ears of Corn: A Guide to People-Centered Agricultural Improvement, World Neighbors, Oklahoma City, OK (1982). This book has been translated into 10 languages. Roland took initiative to introduce SRI in West Timor province of Indonesia and also boosted it elsewhere.
[22] Ana Primavesi, Manejo Ecológico do Solo, 1st edition, Nobel, Sao Paulo, Brazil, Table 3.5, p. 94 (1980).
[23] This book was cited in endnote 13 above. Dr. DeDutta, an agronomist, was for 27 years a principal scientist at the International Rice Research Institute in the Philippines. His book on rice science has been described in Wikipedia as “an authoritative opus in the field of rice cultivation.”
[24] This response reflects Green Revolution scientists’ lack of attention to roots. While writing this paragraph, I ‘googled’ the words Green Revolution and roots. I found that the first 15 items listed were mostly about how paying attention to roots should be part of a next Green Revolution, e.g., J.P. Lynch, ‘Roots of the Second Green Revolution,’ Turner Review: Australian Journal of Botany, 55: 493-512 (2007); G. Den Herder, G. Van Isterdael, T. Beeckman and I. De Smet, ‘The roots of a new green revolution,’ Trends in Plant Science, 15: 600-607 (2010); V. Gewin, ‘Food: An underground revolution,’ Nature, 466: 552-553. This confirmed that Green Revolution research and reporting were focused almost entirely above-ground.
[25] I opened the presentation by thanking Bill Clinton, the US president at the time, who was in the middle of an impeachment process in our Congress. His political troubles were making clear I thought to everyone the difference between an indictment (impeachment) and a conviction (the actual verdict). My presentation, I said would be like the first, making a case for taking SRI seriously. The second, a final judgment, was still to be determined, I hoped with IRRI cooperation. I can’t know how many in audience understood this distinction, probably more important to an American than to others, and to a political scientist more than to agricultural researchers.
[26] M.C. Drew, C.J. He and P.W. Morgan, ‘Programmed cell death and aerenchyma formation in roots,’ Trends in Plant Science, 5: 123-127 (2000).
[27] H.J. Kronzucker, M.Y. Siddiqui, A.D.M. Glass, and G.J.D. Kirk, ‘Nitrate-ammonium synergism in rice: A subcellular flux analysis,’ Plant Physiology, 119:1041-1045 (1999).
[28] G.J.D. Kirk and D. R. Bouldin, ‘Speculations on the operation of the rice root system in relation to nutrient uptake,’ p. 199, in F. W. T. Penning de Vries et al., eds., Simulation and Systems Analysis for Rice Production, Pudoc, Wageningen, Netherlands (1991).
[29] J.K. Ladha, G.J.D. Kirk, J. Bennett, S. Peng, C.K. Reddy and U. Singh, ‘Opportunities for increased nitrogen-use efficiency from improved lowland rice germplasm, Field Crops Research, 56:41-71 (1998).
[30] See p. 197 in the paper cited in endnote 27.
[31] See reference cited in endnote 13. An evaluation of SRI methods done in Japan found the ratios between whitish and blackened roots (alive vs. decaying) to be 21:10 for SRI-grown plants and 10:13 for conventionally-grown ones. T. Chapagain, A. Reisman and E. Yamaji, ‘Assessment of System of Rice Intensification (SRI) and conventional practices under organic and inorganic management in Japan,’ Rice Science, 18: 311-320 (2011).
[32] Anil K. Verma, ‘SRI in Bihar: From one to 350,000,’ Farming Matters, 29: 44-46 (2013).
PICTURE CREDITS: Shuichi Sato (Nippon Koei); Bahman Larijani (Iran); Khidhir Hameed (Iraq).